Even after the war ended, Bohr still called for peace in the uses of atomic energy.

Here, the first atomic bomb was being created, and Bohr raised concerns about the bomb’s potential uses.
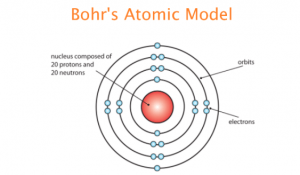
Bohr and one of his sons traveled to the U.S., where Bohr worked with the Manhattan Project in Los Alamos, New Mexico. Eventually, Bohr and his family had to flee Copenhagen as well. Though he retained this title for the rest of his life, the impact of World War II would pull Bohr away from the physical position.īohr was known for his compassion during wartime, donating his Nobel Prize to Finnish war efforts and providing refuge to German-Jewish physicists at the Institute in Copenhagen as Nazi occupation spread. In 1916, he was appointed professor of theoretical physics at the University of Copenhagen, and in 1920, he became head of their Institute for Theoretical Physics. World War II and the Manhattan Projectįrom 1913 to 1916, Niels Bohr held lectureships in physics at the University of Copenhagen and the Victoria University in Manchester. This theory would become crucial in early attempts at splitting uranium atoms, which in turn were an important element in developing the atomic bomb. Bohr presented his liquid droplet theory, which stated that a liquid drop accurately represents an atomic nucleus. In the late 1930s, Bohr collaborated with a group of scientists working on the nuclear fission. This concept emphasized that experimental results are directly impacted by the tools used to achieve them. Understanding of electron properties needs to have a foundation in empirical measurement.An electron can be viewed either as a particle or wave.In his published essays from 1933 to 1962, Bohr formulated a concept called complementarity, which forms the basis of early quantum theory. The atomic model is regarded as the top contribution Bohr made to physics, but it was not his only major contribution. Over the years, Bohr’s base idea was proved to be correct, and he received the Nobel Prize in Physics in 1922. Licensed under CC BY-SA 3.0, via Wikimedia Commons.Īlthough the theory wasn’t perfect, the Bohr atomic model was a better fit for experimental evidence from other physicists. This explains why atoms emit light in fixed wavelengths.Īn animation of Bohr’s model of the atom, including an electron making quantum leaps. The electrons would move from one orbit to another with lower energy, producing a light quantum. In addition, the number of electrons in the outer orbit determine the element’s properties (some of which greatly help to organize the elements as they are discovered). In a three-part series, he wrote that the electrons travel in separate orbits around a small, positively charged nucleus. Bohr challenged the structure theorized by Rutherford and his collaborators, going on to develop the Bohr model of the atom by adapting Rutherford’s structure based on quantum mechanics. His research eventually led him to a close study of the structure of atoms on the basis of Rutherford’s discovery of the atomic nucleus. In 1912, Niels Bohr worked at Ernest Rutherford’s laboratory at Manchester University. This award was the first recognition of Bohr’s soon-to-be prolific career in theoretical physics. For this work, Bohr received a gold medal, and his work was published in the Philosophical Transactions of the Royal Society (1908). This prompted him to produce an experimental and theoretical investigation of surface tension via oscillating fluid jets. This work is in the public domain in its country of origin and other countries and areas where the copyright term is the author’s life plus 70 years or less, via Wikimedia Commons.ĭuring Bohr’s collegiate education, the Academy of Sciences offered a prize for the solution to a specific scientific problem. However, before he had any degree, Bohr demonstrated an extreme competence in physics.Ī photograph of Niels Bohr. Bohr took an early interest in physics, later pursuing his master’s and doctorate degrees at the University of Copenhagen. His family background set him up well for a career in science: His mother came from a family distinguished in education and his father was a well-known physiologist. Niels Bohr was born on October 7, 1885, in Copenhagen, Denmark. Niels Bohr’s Introduction to Theoretical Physics
As a philosopher and physicist, Bohr was a great supporter of scientific research, with his own research bringing us the Bohr model of the atom and the liquid droplet theory. He made great steps forward in understanding quantum theory and atomic structure, the latter of which earned him the Nobel Prize in Physics in 1922. Danish physicist Niels Bohr is one of the most prominent physicists of the 20 th century.
